Systemic defense signaling in Austrian pine
Soumya K Ghosh, Jelmer W Poelstra, David Mackey, Pierluigi Bonello
New Phytol.; 2026 May; 250(4):2495-2515. doi: 10.1111/nph.70866.
Abstract
In plants, the induction of systemic defenses is essential for sustaining structural and physiological homeostasis during repeated attacks by various pests and pathogens. Plant hormones regulate numerous systemic responses; however, relatively little is known about their role in the expression of systemic induced resistance (SIR) against necrotrophic fungi, particularly in trees. Based on an established pathosystem comprised of Austrian pine and Diplodia pinea, we examined the timing of local and systemic accumulation of plant growth and stress hormones and systemic accumulation of transcripts, including those encoding proteins associated with hormone production, signal propagation, and local and systemic defense responses. Differentially expressed genes were enriched for those involved in pattern recognition, defense, and various transcription factors related to jasmonic acid (JA), abscisic acid (ABA), auxin, and gibberellin signaling pathways. The spatiotemporal accumulation of hormones and their precursors and conjugates, including ABA, JA, methyl jasmonate (MeJA), jasmonoyl isoleucine, and 12-oxophytodieonic acid, aligned with the transcriptomic data. Collectively, our results indicate that early ABA signaling and sustained JA signaling, along with mobile MeJA signaling, mediate the full activation of SIR following infection of Austrian pine by D. pinea.
See https://pubmed.ncbi.nlm.nih.gov/41742326/ or
https://nph.onlinelibrary.wiley.com/doi/10.1111/nph.70866
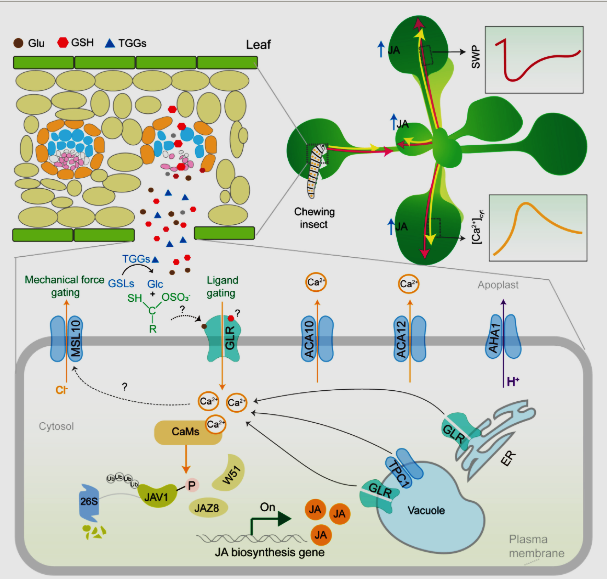
Model for wound-induced electrical and calcium-based defense signaling in Arabidopsis. Mechanical wounding or insect herbivory triggers the release of chemical elicitors (e.g. glutamate (Glu), glutathione (GSH), and β-thioglucoside glucohydrolase (TGGs)) from damaged cells into the apoplast. Extracellular Glu and GSH are likely to bind to the different domains of GLU-like receptor (GLR) proteins for activation of ion channel activities, leading to a transient increase in cytosolic Ca2+ concentrations. In parallel, TGGs hydrolyze glucosinolates to generate unstable aglucone intermediates, which trigger membrane potential changes and [Ca2+]cyt increases. Concurrently, wound-induced turgor pressure changes alter membrane tension and activate mechanosensitive channel of small conductance-like 10, causing anion (e.g. Cl−) efflux and attenuating the magnitude of membrane depolarization. The Ca2+ signal is further amplified by endomembrane-resident GLRs and the tonoplast-localized Two Pore Channel 1, which mediate Ca2+ release from intracellular stores. The wound-induced Ca2+ transients would then activate calmodulin-dependent phosphorylation of jasmonate (JA)-associated valine-glutamine (VQ) motif gene 1 (JAV1) for disintegration of the JAV1-JAZ8-WRKY51 complex, thereby giving rise to the rapid burst of JA for both local and systemic plant defense responses. Finally, the plasma membrane H+-ATPase AHA1 restores membrane potential by extruding protons, while the Ca2+-ATPases ACA10/ACA12 export cytosolic Ca2+ to the apoplast, collectively resetting [Ca2+]cyt to baseline levels. AHA1, H+-ATPase 1; JAZ8, JA-ZIM-DOMAIN PROTEIN 8: ACA – auto-inhibited Ca2+-ATPase. Solid arrows denote experimentally verified pathways. Dashed arrows signify inferred connections requiring experimental validation. CaMS, calmodulin; ER, endoplasmic reticulum; SWP, slow wave potential. (Li et al. 2025)
Views: 29


