|
Synthesis and transfer of galactolipids in the chloroplast envelope membranes of Arabidopsis thaliana
Saturday, 2016/09/24 | 06:01:08
|
|
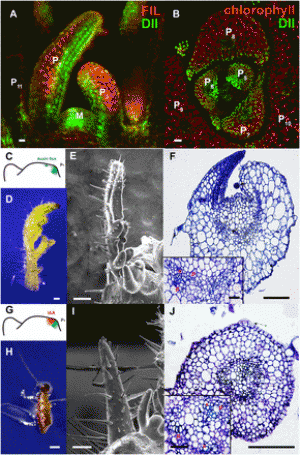
Amélie A. Kelly, Barbara Kalisch, Georg Hölzl, Sandra Schulze, Juliane Thiele, Michael Melzer, Rebecca L. Roston, Christoph Benning, and Peter Dörmann SignificanceEstablishment of the progenitor of chloroplasts by the host plant cell during endosymbiosis required the integration of two sets of biological membranes, the endoplasmic reticulum and the chloroplast envelopes, participating in the synthesis of galactolipid precursors for the photosynthetic membranes. Galactolipid synthesis is unequally distributed between the two envelope membranes, necessitating lipid transfer between the envelopes and toward the thylakoids. Here we show that the N-terminal sequence of digalactosyldiacylglycerol synthase 1 is essential for the integration of the chloroplast galactolipid synthesis machinery into the host cell. This N-terminal sequence was invented at the time the endosymbiotic organelle was established, providing a basic glycosyltransferase with a neofunction essential for lipid mobilization between organelles and endomembrane systems in plants. AbstractGalactolipids [monogalactosyldiacylglycerol (MGDG) and digalactosyldiacylglycerol (DGDG)] are the hallmark lipids of photosynthetic membranes. The galactolipid synthases MGD1 and DGD1 catalyze consecutive galactosyltransfer reactions but localize to the inner and outer chloroplast envelopes, respectively, necessitating intermembrane lipid transfer. Here we show that the N-terminal sequence of DGD1 (NDGD1) is required for galactolipid transfer between the envelopes. Different diglycosyllipid synthases (DGD1, DGD2, and Chloroflexus glucosyltransferase) were introduced into the dgd1-1 mutant of Arabidopsis in fusion with N-terminal extensions (NDGD1 and NDGD2) targeting to the outer envelope. Reconstruction of DGDG synthesis in the outer envelope membrane was observed only with diglycosyllipid synthase fusion proteins carrying NDGD1, indicating that NDGD1 enables galactolipid translocation between envelopes. NDGD1 binds to phosphatidic acid (PA) in membranes and mediates PA-dependent membrane fusion in vitro. These findings provide a mechanism for the sorting and selective channeling of lipid precursors between the galactolipid pools of the two envelope membranes.
See: http://www.pnas.org/content/113/38/10714.abstract.html?etoc PNAS September 20 2016; vol.113; no.38: 10714–10719
Fig. 2. NDGD1 interactions with lipids and membranes. (A, Upper) NDGD1 binds to PA as revealed after incubation of nitrocellulose strips containing different glycerolipids with recombinant Nus (control), DGD1, or NDGD1 protein. Binding was visualized by immunodetection. (Lower) The blot shows NDGD1 binding to different amounts of PA (0.1–10 nmol). CL, cardiolipin; DAG, diacylglycerol; PC, phosphatidylcholine; PE, phosphatidylethanolamine; PG, phosphatidylglycerol. (B) NDGD1 binding to liposomes is PA-dependent. Liposomes with different proportions of PA and PC were incubated with recombinant proteins. Bound proteins were detected in polyacrylamide gels after centrifugation of liposomes. (Upper Left) Control (Nus) protein; P, pellet, S, supernatant. (Upper Right) NDGD1 binding to liposomes composed of PA and PC. Composition is expressed as percent PA. (Lower) Binding of NDGD1 (2–10 µg) to liposomes consisting of 60% PC/40% PA or 100% PC. |
|
|
|
[ Other News ]___________________________________________________
|


 Curently online :
Curently online :
 Total visitors :
Total visitors :
(26).png)